Advertisements
Advertisements
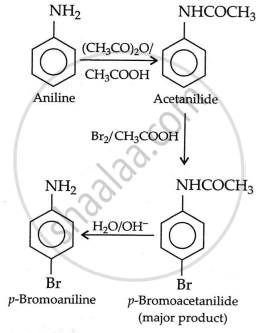
प्रश्न
Accomplish the following conversion:
Aniline to p-bromoaniline
उत्तर
APPEARS IN
संबंधित प्रश्न
Identify the weakest base amongst the following :
(a) p- methoxyaniline
(b) o-toluidine
(c) benzene - 1, 4 - diamine
(d) 4 - aminobenzoic acid
How will you convert Benzene into aniline?
Arrange the following in increasing order of their basic strength:
C2H5NH2, C6H5NH2, NH3, C6H5CH2NH2 and (C2H5)2NH
Account for the following:
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Complete the following reactions:
`C_6H_5N_2Cl+H_3PO_2+H_2O->`
Give reason (CH3)2NH is more basic than (CH3)3N in an aqueous solution.
Illustrate the following reactions giving suitable example in each case
Ammonolysis
Arrange the following in increasing order of basic strength :
C6H5NH2, C6H5NHCH3, C6H5N(CH3)2
Answer in one sentence.
Arrange the following amines in increasing order of boiling points.
n-propylamine, ethylmethyl amine, trimethylamine.
Arrange the following compounds in increasing order of their boiling points.
Ethyl alcohol, Ethyl amine, Ethanoic acid, Ethane
Among the following isomeric amines, an amine having highest boiling point is:
Assertion: N-Ethylbenzene sulphonamide is soluble in alkali.
Reason: Hydrogen attached to nitrogen in sulphonamide is strongly acidic.
Acetic acid exist as dimer in benzene due to
Dimerisation of carboxylic acids is due to
Arrange the following in increasing order of their boiling point:
C2H5OH, C2H5NH2, (C2H5)3N
Arrange the decreasing boiling point.
\[\ce{CH3COOH, C2H5OH, CH3NH2, CH3OCH3}\]
