Advertisements
Advertisements
प्रश्न
Write a short note on the following:
Coupling reaction
उत्तर
The azo products obtained have an extended conjugate system having both the aromatic rings joined through the –N=N– bond. These compounds are often coloured and are used as dyes. Benzene diazonium chloride reacts with phenol in which the phenol molecule at its para position is coupled with the diazonium salt to form p-hydroxyazobenzene. This type of reaction is known as a coupling reaction. Similarly, the reaction of diazonium salt with aniline yields p-aminoazobenzene. This is an example of an electrophilic substitution reaction.
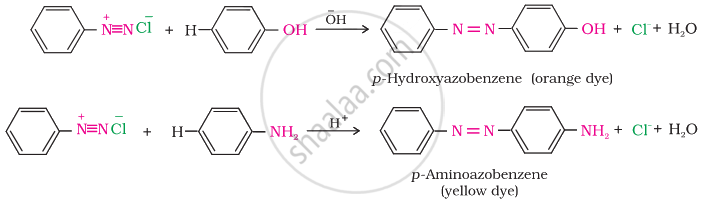
APPEARS IN
संबंधित प्रश्न
Illustrate the following reactions giving suitable example in each case
Coupling reaction
Write equations of the following reactions:
Coupling reaction
Which of the following compound will not undergo azo coupling reaction with benzene diazonium chloride?
The product of the following reaction is:
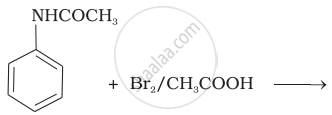
(i)
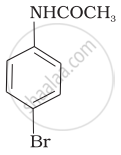
(ii)
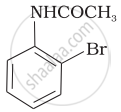
(iii)
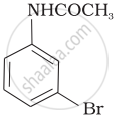
(iv)
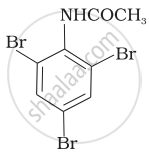
Which of the following reactions belong to electrophilic aromatic substitution?
(i) Bromination of acetanilide
(ii) Coupling reaction of aryldiazonium salts
(iii) Diazotisation of aniline
(iv) Acylation of aniline
Under what reaction conditions (acidic/basic), the coupling reaction of aryldiazonium chloride with aniline is carried out?
Benzenediazonium chloride reacts with phenol to give p-hydroxy azobenzene, an orange dye. This reaction is known as ______.
When benzene diazonium chloride reacts with phenol, it forms a dye. This reaction is called ______.
