Advertisements
Advertisements
The energy released when an electron is added to a neutral gaseous isolated atom to form a negatively charged iron.
Concept: Periodic Properties > Electron Affinity
A process of formation of ions from molecules which are not in the ionic state
Concept: Periodic Properties > Ionisation Potential (Ionisation Energy)
Give a reason for Ionisation potential increases across a period, from left to right
Concept: Periodic Properties > Ionisation Potential (Ionisation Energy)
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element B would have ______ metallic character than A.
Concept: Periodic Properties > Metallic and Non-metallic Characters
Define the term Electronegativity.
Concept: Periodic Properties > Electronegativity
An element in period 3, whose electron affinity is zero:
Concept: Periodic Properties > Electron Affinity
The most electronegative element from the following elements is:
Concept: Periodic Properties > Electronegativity
In Period 3, the most metallic element is ______.
Concept: Periodic Properties > Metallic and Non-metallic Characters
K, Pb, Ca, Zn (In the increasing order of the reactivity)
Concept: Periodic Properties
Mg2+, Cu2+, Na1+, H1+ (In the order of preferential discharge at the cathode)
Concept: Periodic Properties > Metallic and Non-metallic Characters
Li, K, Na, H (In the decreasing order of their ionization potential)
Concept: Periodic Properties > Ionisation Potential (Ionisation Energy)
F, B, N, O (In the increasing order of electron affinity)
Concept: Periodic Properties > Electron Affinity
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 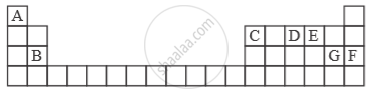
Which element forms electrovalent compound with G?
Concept: Periodic Properties > Electronegativity
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 
The ion of which element will migrate towards the cathode during electrolysis?
Concept: Periodic Properties
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 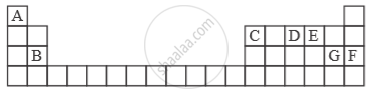
Which non-metallic element has the valency of 2?
Concept: Periodic Properties > Metallic and Non-metallic Characters
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 
Which is an inert gas?
Concept: Periodic Properties
An element with the largest atomic radius among the following is ______.
Concept: Periodic Properties > Atomic Radius Or Atomic Size
Arrange the following as per the instruction given in the bracket:
P, Si, F, Be (decreasing order of valence electrons)
Concept: Periodic Properties > Valency
Give reason for the following:
Ionisation potential decreases down a group.
Concept: Periodic Properties > Ionisation Potential (Ionisation Energy)
In the 2nd period Neon has maximum Ionization Potential because ______.
Concept: Periodic Properties > Shells (Orbits)
