Advertisements
Advertisements
Question
Phenoxide ion is more stable than phenol due to the ____________.
Options
resonating structures of benzene ring
delocalisation of positive charge in phenoxide ion
delocalisation of negative charge in phenoxide ion
negative charge on oxygen atom
MCQ
Fill in the Blanks
Solution
Phenoxide ion is more stable than phenol due to the delocalisation of negative charge in phenoxide ion.
Explanation:
Due to delocalisation of negative charge, phenoxide ion is more stable than phenol.
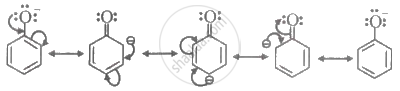
shaalaa.com
Alcohols and Phenols
Is there an error in this question or solution?
