Advertisements
Advertisements
प्रश्न
2 mL of sodium hydroxide solution is added to a few pieces of granulated zinc metal taken in a test tube. When the contents are warmed, a gas evolves which is bubbled through a soap solution before testing. Write the equation of the chemical reaction involved and the test to detect the gas. Name the gas which will be evolved when the same metal reacts with dilute solution of a strong acid.
उत्तर
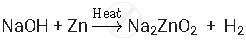
Test for hydrogen: A splint is lit and held near the opening of the tube, then the stopper is removed to expose the splint to the gas. If the gas is flammable, the mixture ignites. This test is most commonly used to identify hydrogen, which ignites with a distinctive 'squeaky pop' sound
Zn + H2SO4 (dil) → ZnSO4 + H2
APPEARS IN
संबंधित प्रश्न
Translate the following statement into chemical equation and then balance it.
Hydrogen sulphide gas burns in the air to give water and sulphur dioxide.
Balance the given chemical equation:
NH3 → N2 + H2
Is Respiration an endothermic reaction or an exothermic reaction?
Write your observations and name the product when
Iron nails are added to an aqueous solution of copper sulphate.
To determine the percentage of water absorbed by raisins, raisins are soaked in water for:
What is a chemical equation? Why it is necessary to balance it?
Balance the following equation:
P + HNO3 → NO2 + H2O + H3PO4
Write the balanced chemical equation of the following reaction.
aluminium carbide + water → aluminium hydroxide + methane
Answer the following question.
What is observed when 2 mL of dilute hydrochloric acid is added to 1 g of sodium carbonate taken in a clean and dry test tube? Write a chemical equation for the reaction involved.
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
C2H5OH + 3O2 → 2CO2 + 2H2O
