Advertisements
Advertisements
प्रश्न
| A covalent bond in tert-butyl bromide breaks in a suitable polar solvent to give ions. |
- Name the anion produced by this breaking of a covalent bond.
- Indicate the type of bond breaking in this case.
- Comment on the geometry of the cation formed by such bond cleavage.
उत्तर
- \[\begin{array}{cc}
\ce{CH3}\phantom{..............}\ce{CH3}\phantom{.........}\\
|\phantom{..................}|\phantom{............}\\
\ce{H3C - C - Br} \ce{->[aq]} \ce{H3C - C^\oplus} + \ce{\underset{Bromide ion (anion)}{Br-}}\\
|\phantom{..................}|\phantom{............}\\
\ce{CH3}\phantom{..........}\ce{\underset{tert-butyl Cation}{CH3}}\phantom{.....}\\
\end{array}\] - Heterolytic cleavage/fission takes place as charged ions are produced.
- tert-Butyl carbocation formed in the given cleavage has trigonal planar geometry.
APPEARS IN
संबंधित प्रश्न
Identify a more favourable resonance structure from the following. Justify.

Match the pairs.
| Column 'A' | Column 'B' | ||
| i. | Inductive effect | a. | delocalisation of π electrons |
| ii. | Hyperconjugation | b. | displacement of π electrons |
| iii. | Resonance effect | c. | delocalisation of σ electrons |
| d. | displacement of σ electrons | ||
Write IUPAC names of the following.

Write the IUPAC name of the following.

Observe the following structures and answer the questions given below.
- \[\ce{CH3 - CH2 - CH2 - CHO}\]
\[\begin{array}{cc}\ce{CH3 - CH - CHO}\\
|\phantom{...}\\\ce{CH3}\end{array}\]
a. What is the relation between (i) and (ii)?
b. Write IUPAC name of (ii).
c. Draw the functional group isomer of (i).
Phytane is a naturally occurring alkane produced by the alga spirogyra and is a constituent of petroleum. The IUPAC name for phytane is 2,6,10,14-tetramethylhexadecane. Write a zig-zag formula for phytane. How many primary, secondary, tertiary, and quaternary carbons are present in this molecule?
The IUPAC name of \[\begin{array}{cc}
\phantom{.}\ce{CH3}\\|\phantom{..}\\
\ce{H3C - C - CH = C(CH3)2}\\
|\phantom{..}\\\phantom{..}\ce{CH3}
\end{array}\] is
What is meant by a functional group?
Give the IUPAC names of the following compound.
\[\ce{(CH3)2 CH–CH2 –CH(CH3 )–CH(CH3)2}\]
Give the IUPAC names of the following compound.
\[\ce{CH3 - O - CH3}\]
Give the IUPAC names of the following compound.
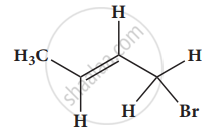
Give the IUPAC names of the following compound.
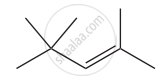
Give the IUPAC names of the following compound.
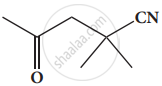
Give the IUPAC names of the following compound.

Give the structure for the following compound.
3 - Chlorobutanal
Give the structure for the following compound.
3 - Chlorobutanol
Give the structure for the following compound.
2,2-dimethyl-1-chloropropane
Give the structure for the following compound.
Butan - 2, 2 - diol
Give the structure for the following compound.
3-Chlorobut - 1 - ene
As per IUPAC nomenclature, the name of the complex [Fe(H2O)5(NCS)]2+ is ____________.
Hybridisation of C-atom to which -OH group is attached in the below given compound is:
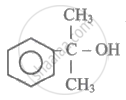
As per IUPAC nomenclature, the name of the complex Na3[AlF6] is ____________.
IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3 - C - CH2CH3}\\
|\phantom{....}\\
\ce{OH}\phantom{..}
\end{array}\]
The IUPAC name of the following compound is:
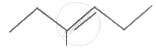
The IUPAC name of the following compound is:
CH2 = C = CH − CH3
The correct structure of 2,6-Dimethyl-dec-4-ene is ______
The IUPAC name of the following compound is:
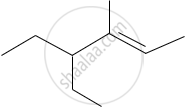
Identify the α-carbons in the following species and give the total number of α-hydrogen.
CH2 = CH - CH2 - CH3
