Advertisements
Advertisements
प्रश्न
A reaction, A + B → C + D + q is found to have a positive entropy change. The reaction will be ______.
विकल्प
possible at high temperature
possible only at low temperature
not possible at any temperature
possible at any temperature
उत्तर
A reaction, A + B → C + D + q is found to have a positive entropy change. The reaction will be possible at any temperature.
Explanation:
For a reaction to be spontaneous, ΔG should be negative.
ΔG = ΔH – TΔS
According to the question, for the given reaction,
ΔS = positive
ΔH = negative (since heat is evolved)
⇒ ΔG = negative
Therefore, the reaction is spontaneous at any temperature.
APPEARS IN
संबंधित प्रश्न
For the reaction, \[\ce{2Cl_{(g)} → Cl_{2(g)}}\], what are the signs of ΔH and ΔS?
The entropy change can be calculated by using the expression ∆S = `q_(rev)/T`. When water freezes in a glass beaker, choose the correct statement amongst the following :
The spontaneity means, having the potential to proceed without the assistance of external agency. The processes which occur spontaneously are:
(i) Flow of heat from colder to warmer body.
(ii) Gas in a container contracting into one corner.
(iii) Gas expanding to fill the available volume.
(iv) Burning carbon in oxygen to give carbon dioxide.
Enthalpy diagram for a particular reaction is given in figure. Is it possible to decide spontaneity of a reaction from given diagram. Explain.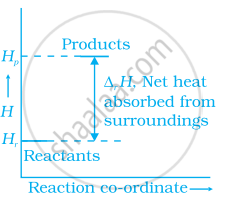
Match the following processes with entropy change:
| Reaction | Entropy change |
| (i) A liquid vapourises | (a) ∆S = 0 |
| (ii) Reaction is non-spontaneous at all temperatures and ∆H is positive |
(b) ∆S = positive |
| (iii) Reversible expansion of an ideal gas | (c) ∆S = negative |
Assertion (A): A liquid crystallises into a solid and is accompanied by decrease in entropy.
Reason (R): In crystals, molecules organise in an ordered manner.
For a sample of perfect gas when its pressure is changed isothermally from Pi to Pf, the entropy change is given by ______.
For the irreversible expansion of an ideal gas under isothermal conditions, the correct option is ______.
The process with negative entropy change is ______.
A reaction at 1 bar is non-spontaneous at low temperatures but becomes spontaneous at high temperatures. Identify the correct statement about the reaction among the following.
