Advertisements
Advertisements
प्रश्न
A salt X is formed and a gas is evolved when ethanoic acid reacts with sodium hydrogencarbonate. Name the salt X and the gas evolved. Describe an activity and draw the diagram of the apparatus to prove that the evolved gas is the one which you have named. Also, write chemical equation of the reaction involved.
उत्तर
When ethanoic acid reacts with sodium hydrogencarbonate, it gives rise to a salt (sodium acetate), carbon dioxide and water.
`underset("(ethanoic acid)")("CH"_3"COOH") + underset("sodium hydrogencarbonate")("NaHCO"_3) -> underset("Sodium acetate")("CH"_3"COONa") + "H"_2"O" + underset("(Carbon dioxide)")("CO"_2)`
Hence, the salt X is sodium acetate and the gas is carbon dioxide.
Activity:
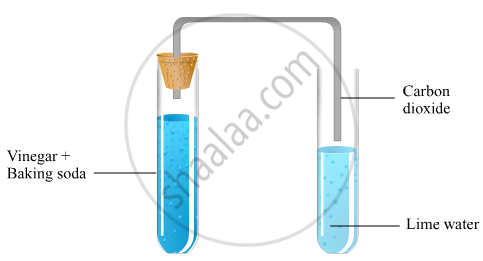
On passing this evolved gas through freshly prepared lime water, calcium carbonate is formed which makes lime water milky.
`"CO"_2 + "Ca"("OH")_2 -> "CaCO"_3 + "H"_2"O"`
The turning of lime water into milky is a standard test of carbon dioxide.
