Advertisements
Advertisements
प्रश्न
According to the valence bond theory, the hybridization of central metal atom is dsp2 for which one of the following compounds?
विकल्प
\[\ce{Na2[NiCl4]}\]
\[\ce{NiCl2 * 6H2O}\]
\[\ce{K2[Ni(CN)4]}\]
\[\ce{[Ni(CO)4]}\]
उत्तर
\[\ce{K2[Ni(CN)4]}\]
Explanation:
We must first determine the coordination number of the core metal ion in order to find the complex that contains the central metal atom with dsp-2 hybridization. From there, we can determine that the main metal ion in the coordination complex has hybridised.
In \[\ce{Na2[NiCl_4]}\], Ni exists in +2 oxidation state. The `"Cl"^-` is a weak field ligand. The coordination number of Ni is 4 hence, it undergoes `sp^3` hybridization.
For \[\ce{NiCl2 * 6H2O}\]
| Ni | [Ar]3d8 4s2 |
| Ni2+ | [Ar] 3d8 4s0 |

In \[\ce{NiCl2 * 6H2O}\], Ni occurs in +2 oxidation state. The coordination number of Ni is 6.
As H2O is a weak ligand, Ni+2 undergoes sp3d2 hybridisation as follows:
| Ni | [Ar]3d8 4s2 |
| Ni2+ | [Ar] 3d8 4s0 |
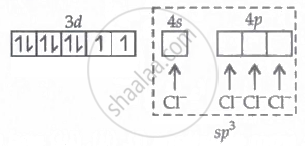
In K2[Ni(CN)4], Ni occurs in +2 oxidation state. The CN- is a strong field ligand. The Ni undergoes dsp2 hybridization as shown below.

In Ni(CO)4, the Ni atom undergoes sp3 hybridization as shown below.
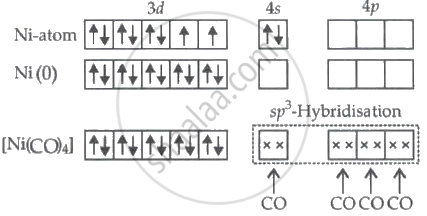
Therefore, according to valence bond theory, the hybridization of central metal atom is dsp2 for K2[Ni(CN)4].
