Advertisements
Advertisements
प्रश्न
Account for the following:
Carboxylic acid is a stronger acid than phenol.
उत्तर
Carboxylic acids are acidic due to resonance stabilisation of carboxylate anion and in phenols, acidic character is present due to resonance stabilisation of phenoxide anion. Carboxylic acids are more acidic than phenols because the negative charge in carboxylate anion is more spread out compared to the phenoxide ion, as there are two electronegative O-atoms in carboxylate anion compared to one in phenoxide ion. In the resonance structures of carboxylate anion, the negative charge is present on the O-atoms, while in resonance of phenoxide ion, negative charge is also present on electropositive carbon atom, which leads to less stability of phenoxide ion than carboxylate anion.
APPEARS IN
संबंधित प्रश्न
How is 1-nitropropane prepared from suitable oxime?
How are 2 - nitropropane prepared from suitable oxime?
CH3CO2H or CH2FCO2H
Which acid of the pair shown here would you expect to be stronger?
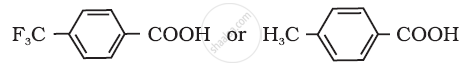
Arrange the following compounds in increasing order of their property as indicated:
Benzoic acid, 4-Nitrobenzoic acid, 3, 4-Dinitrobenzoic acid, 4-Methoxybenzoic acid (acid strength)
Assertion: Formaldehyde is a planar molecule.
Reason: It contains sp2 hybridised carbon atom.
A mixture of benzaldehyde and formaldehyde on heating with 50% NaOH solution gives
Formic acid and formaldehyde can be distinguished by treating with ______.
Describe the action of alcoholic potassium hydroxide (alc. KOH) on ethyl bromide
Describe the action of alcoholic potassium hydroxide (alc. KOH) on n-propyl bromide
