Advertisements
Advertisements
प्रश्न
Amines are basic in nature. The pKb value is a measure of the basic strength of an amine. Lower the value of pKb, more basic is the amine. The effect of substituent on the basic strength of amines in aqueous solution was determined using titrations. The substituent “X” replaced \[\ce{^”-CH2^“}\] group in piperidine (compound 1) and propylamine \[\ce{CH3CH2CH2NH2}\], (compound 2).
Compound 1: 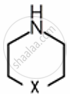
Compound 2: \[\ce{HXCH2CH2NH2}\]
The experimental data is tabulated below:
| Substituent “X” | Electro-negativity of X | Substituted piperidine compound | pKa | Substituted propylamine compound | pKa |
| \[\ce{CH2}\] | 2.55 | 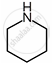 |
11.13 | \[\ce{CH3CH2CH2NH2}\] | 10.67 |
| \[\ce{NH}\] | 3.12 | 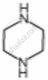 |
9.81 | \[\ce{NH2CH2CH2NH2}\] | 10.08 |
| \[\ce{O}\] | 3.44 | 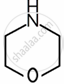 |
8.36 | \[\ce{HOCH2CH2NH2}\] | 9.45 |
| \[\ce{CH3CON}\] | 3.6 | 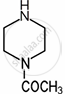 |
7.94 | \[\ce{CH3CONHCH2CH2NH2}\] | 9.28 |
| \[\ce{C6H5CON}\] | 3.7 | 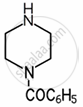 |
7.78 | \[\ce{C6H5CONHCH2CH2NH2}\] | ______ |
Study the above data and answer the following questions:
a. Plot a graph between the electro-negativity of the substituent vs. the pKb value of the corresponding substituted propyl amine (given that pKa + pKb = 14). Is there any relation between the electro-negativity of the substituent and its basic strength?
b. The electronegativity of the substituent \[\ce{^“C6H5CON^“}\] is 3.7; what is the expected pKa value of compound \[\ce{C6H5CONHCH2CH2NH2}\]?
- 9.9
- 9.5
- 9.3
- 9.1
c. The pKa value of the substituted piperidine formed with the substituent “X” is found to be 8.28. What is the expected electronegativity of “X”?
- 3.5
- 3.4
- 3.8
- 3.1
OR
What is the most suitable pKa value of the substituted propylamine formed with substituent “X” with electronegativity 3.0?
- 10.67
- 10.08
- 10.15
- 11.10
उत्तर
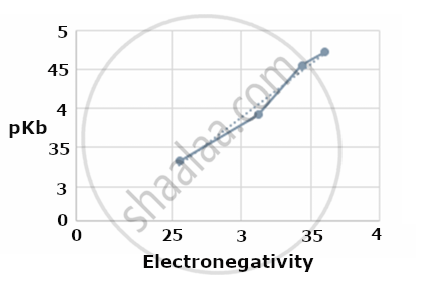
The pKb increases with an increase in the electronegativity of the substituent; therefore, the basic strength decreases with an increase in the electronegativity of the substituent- 9.1
- 3.5
OR
10.15
