Advertisements
Advertisements
प्रश्न
Answer in brief.
What are bidentate ligands? Give one example.
उत्तर
Bidentate ligands: The ligands which bind to central metal through two donor atoms are called bidentate ligands.
e.g.
1. Ethylenediammine binds to the central metal atom through two nitrogen atoms.

2. Similarly, Oxalate ligand `"C"_2"O"_4^{2Θ}` utilizes electron pair on each of its negatively charged oxygen atoms on linking with central metal.
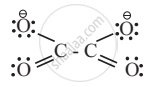
APPEARS IN
संबंधित प्रश्न
What is meant by the chelate effect?
Answer in brief.
What are the coordination number and oxidation state of the metal ion in the complex [Pt(NH3)Cl5]-?
Answer in brief.
What is the difference between a double salt and a complex? Give an example.
What are ligands?
What are the types of ligands? Give one example of each type.
Amongst the following, the ambidentate ligand is ____________
Write one example of each of bidentate and ambidentate ligand.
Which of the following is an ambidentate ligand?
Which among the following coordination compounds does not have coordination number equal to number of ligands?
Identify monodentate ligand from following.
Explain monodentate and ambidentate ligands with example.
Give the steps to understand the metal-ligand bonding.
Define monodentate ligand.
Write a Example monodentate ligand.
What are ligands?
What are ligands types? Give one example of each type.
What are types of ligands? Give one example of each type.
What are the types of ligands? Give one example of each type.
What are ligands?
What are the types of ligands? Give one example of each type.
What are ligands?
What are ligands?
What are ligands?
Give an example of chelate effect.
