Advertisements
Advertisements
प्रश्न
Arrange the following hydrogen halides in order of their decreasing reactivity with propene.
विकल्प
HCl > HBr > HI
HBr > HI > HCl
HI > HBr > HCl
HCl > HI > HBr
उत्तर
HI > HBr > HCl
Explanation:
The decreasing order of reactivity of hydrogen halides with propene is HI > HBr > HCl. As the size of halogen increases, the strength of H – X bond decreases and hence, reactivity increases.
APPEARS IN
संबंधित प्रश्न
An alkene ‘A’ contains three C – C, eight C – H σ bonds and one C – C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write IUPAC name of ‘A’.
Write a chemical equation for combustion reaction of the following hydrocarbon:
Butane
Write a chemical equation for combustion reaction of the following hydrocarbon:
Hexyne
Write a chemical equation for combustion reaction of the following hydrocarbon:
Toluene
The addition of HBr to 1-butene gives a mixture of products A, B and C
| (A) | 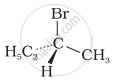 |
| (B) | 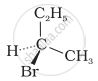 |
| (C) | CH3 – CH2 – CH2 – CH2 – Br |
The mixture consists of:
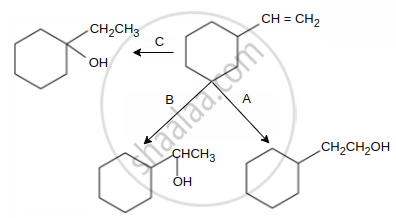
Select schemes A, B, C out of
(I) acid catalysed hydration
(II) HBO
(III) oxymercuration-demercuration
Propene, \[\ce{CH3 - CH = CH2}\] can be converted to 1-propanol by oxidation. Which set of reagents among the following is ideal to effect the conversion ______.
3-Methyl-pent-2-ene on reaction with HBr in presence of peroxide forms an addition product. The number of possible stereoisomers for the product is ______.
In the presence of peroxide, HCl and HI do not give anti-Markovnikov's addition of alkenes because ______.
An alkene ‘A’ contains three C – C, eight C – H σ bonds and one C – C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write the IUPAC name of ‘A’.
