Advertisements
Advertisements
प्रश्न
Assertion (A): It is advised that while diluting an acid one should add water to acid and not acid to water keeping the solution continuously stirred.
Reason (R): The process of dissolving an acid into water is highly exothermic.
विकल्प
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of (A).
Both Assertion (A) and Reason (R) are true but Reason (R) is not the correct explanation of (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
उत्तर
Assertion (A) is false, but Reason (R) is true.
Explanation:
It is crucial to add the acid to the water before adding the water to the mixture of acid and water. This is because acid and water react vigorously exothermically, producing heat and occasionally boiling the liquid.
APPEARS IN
संबंधित प्रश्न
In terms of acidic strength, which one of the following is in the correct increasing order?
What is formed when zinc reacts with sodium hydroxide?
Sodium hydroxide is a ____________.
Curd cannot be stored in
(i) Brass vessel
(ii) Copper vessel
(iii) Steel
(iv) Bronze
How will you protect yourself from the heat generated while diluting a concentrated acid?
What will happen if we take dry HCl gas instead of the aqueous solution of HCl?
What will be the action of the following substances on blue litmus paper?
Dry HCl gas, Moistened NH3 gas, Lemon juice, Carbonated soft drink, Curd, Soap solution.
In one of the industrial processes used for the manufacture of sodium hydroxide, a gas X is formed as by product. The gas X reacts with lime water to give a compound Y which is used as a bleaching agent in chemical industry. Identify X and Y giving the chemical equation of the reactions involved.
The change in colour of the moist litmus paper in the given set up is due to
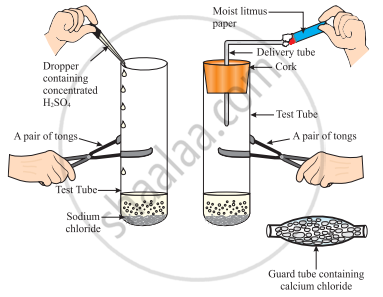
- presence of acid
- presence of base
- presence of H⁺(aq) in the solution
- presence of Litmus which acts as an indicator
Four solutions, namely glucose, alcohol, hydrochloric acid and sulphuric acid filled in four separate beakers are connected one by one in an electric circuit with a bulb. The solutions in which the bulb will glow when current is passed are ______.
