Advertisements
Advertisements
प्रश्न
Assertion: Nitration of chlorobenzene leads to the formation of m-nitrochlorobenzene.
Reason: –NO2 group is a m-directing group.
विकल्प
Assertion and reason both are correct and reason is correct explanation of assertion.
Assertion and reason both are wrong statements.
Assertion is correct but reason is wrong statement.
Assertion is wrong but reason is correct statement.
Assertion and reason both are correct statements but reason is not correct explanation of assertion.
उत्तर
Assertion is wrong but reason is correct statement.
Explanation:
Nitration of chlorobenzene gives ortho and para nitro chlorobenzene. However, nitro group is meta directing in nature.
APPEARS IN
संबंधित प्रश्न
which of the following carbocations is least stable?
(A) 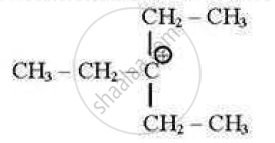
(B)![]()
(C)![]()
(D)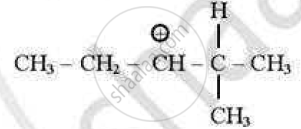
Which one of the following has the highest dipole moment?
C – X bond is strongest in ____________.
Which of the following will have the maximum dipole moment?
Write a test to detect the presence of double bond in a molecule.
Discuss the nature of C – X bond in the haloarenes.
Out of the Chloromethane and Fluoromethane, which one is has higher dipole moment and why?
Why haloarenes are Less reactive than halo alkanes?
