Advertisements
Advertisements
प्रश्न
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
विकल्प
Assertion and reason both are correct and reason is correct explanation of assertion.
Assertion and reason both are wrong statements.
Assertion is correct statement but reason is wrong statement.
Assertion is wrong statement but reason is correct statement.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
उत्तर
Assertion and reason both are wrong statements.
Explanation:
Phenol forms 2, 4, 6-tribromophenol on treatment with bromine in water. In phenols, the polarization of bromine takes place even in the absence of Lewis acid.
APPEARS IN
संबंधित प्रश्न
Name the following compound according to IUPAC system.
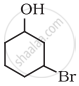
Write IUPAC name of the following compound:
C6H5 – O – C2H5
Give IUPAC name of the following ether:
CH3CH2CH2OCH3
What is the action of hot HI on it?
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
Write the structures of the products when Butan-2-ol reacts with CrO3
What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
Isopropyl alcohol on oxidation forms:
Ethyl alcohol is industrially prepared from ethylene by:
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
