Advertisements
Advertisements
प्रश्न
At least one carbon bond in an unsaturated hydrocarbon is ______.
विकल्प
single
all double
ionic
carbon
give and take
hydrogen
multiple
share
most
covalent
उत्तर
At least one carbon bond in an unsaturated hydrocarbon is multiple.
APPEARS IN
संबंधित प्रश्न
The general molecular formula of alkynes is ______
Biodegradable detergents are made of ______ chain hydrocarbons.
Answer the following question using the data given below:
- Assertion: Alkanes are saturated hydrocarbons.
- Reason: Hydrocarbons consist of covalent bonds.
______ is an odourless and highly inflammable gas.
Hydrocarbons are less dense than ______.
Sewage sludge can also be decomposed by microorganisms to produce methane gas along with impurities like carbon dioxide and ______.
______ can also be used as a refrigerant.
Match the following:
| 1. | Hydrocarbons | a. | Marsh gas |
| 2. | Methane | b. | Cleanest fuel |
| 3. | Butane | c. | Catenation |
| 4. | CNG | d. | Polystyrene |
Write any 2 properties of hydrocarbons.
The hydrocarbons containing carbon-carbon multiple bonds are called ______.
Identify saturated and unsaturated hydrocarbons from the given structural formula:
| \[\begin{array}{cc} \phantom{}\ce{H}\phantom{..}\ce{H}\phantom{}\\ \phantom{}|\phantom{...}|\phantom{}\\ \ce{H-C-C-H}\\ \phantom{}|\phantom{...}|\phantom{}\\ \phantom{}\ce{H}\phantom{..}\ce{H}\phantom{} \end{array}\] |
\[\begin{array}{cc} \phantom{..}\ce{H}\phantom{........}\ce{H}\phantom{.}\\ \phantom{.}\backslash\phantom{......}/\phantom{}\\ \ce{C = C}\\ \phantom{}/\phantom{......}\backslash\phantom{}\\ \phantom{}\ce{H}\phantom{........}\ce{H}\phantom{} \end{array}\] |
| (1) | (2) |
Draw electron dot structure for the following:
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{..}\ce{H}\phantom{}\\
\phantom{}|\phantom{...}|\phantom{}\\
\ce{H-C-C-H}\\
\phantom{}|\phantom{...}|\phantom{}\\
\phantom{}\ce{H}\phantom{..}\ce{H}\phantom{}
\end{array}\]
Draw electron dot structure for the following:
\[\begin{array}{cc}
\phantom{..}\ce{H}\phantom{........}\ce{H}\phantom{.}\\
\phantom{.}\backslash\phantom{......}/\phantom{}\\
\ce{C = C}\\
\phantom{}/\phantom{......}\backslash\phantom{}\\
\phantom{}\ce{H}\phantom{........}\ce{H}\phantom{}
\end{array}\]
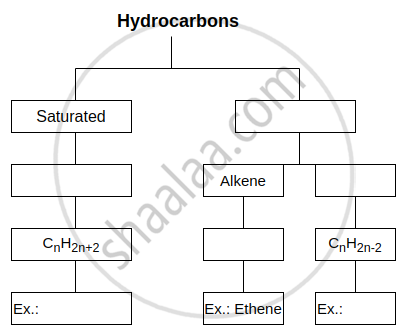
Complete the following flow chart:
Identify the hydrocarbon from the given electron-dot structure:
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{....}\ce{H}\phantom{}\\
\phantom{}\ce{H}\phantom{..}\ce{\text{}^\bullet_\bullet}\phantom{..}\ce{\overset{\bullet\phantom{.}\bullet}{\underset{\bullet\phantom{.}\bullet}{C}}}\phantom{..}\ce{\text{}^\bullet_\bullet}\phantom{..}\ce{\overset{\bullet\phantom{.}\bullet}{\underset{\bullet\phantom{.}\bullet}{C}}}\phantom{..}\ce{\text{}^\bullet_\bullet}\phantom{..}\ce{H}\phantom{}\\
\phantom{}\ce{H}\phantom{....}\ce{H}\phantom{}\\
\end{array}\]
