Advertisements
Advertisements
प्रश्न
Carbonic acid gives an acid salt but hydrochloric acid does not. Explain.
उत्तर
Carbonic acid is a dibasic acid i.e.; H2CO3. So, when it reacts with a base such as NaOH then there is only partial replacement of hydrogen ions of carbonic acid by sodium ion to form an acid salt as:
H2CO3 + NaOH → NaHCO3 + H2O
In case of hydrogen chloride which is a monobasic acid the salt produced will be normal as :
HCl + NaOH → NaCI + H2O
Concept Insight : Salts formed by partial replacement of hydrogen ions of acid by metallic ions are called acid salts.
APPEARS IN
संबंधित प्रश्न
When a solution is added to a cloth strip treated with onion extract, then the smell of onion cannot be detected. State whether the given solution contains an acid or a base.\
Name the gas evolved when dilute HCl reacts with sodium hydrogencarbonate. How is it recognised?
The indicator which produces a pink colour in an alkaline solution is:
(a) methyl orange
(b) turmeric paper
(c) phenolphthalein
(d) litmus paper
Tartaric acid is a ______ acid.
Vinu and Priyan take their lunch at school. Vinu eats lemon rice and Priyan eats curd rice. Both lemon rice and curd rice are sour in taste. What is the reason?
Fill in the crossword given in Figure 5.2 with the help of the clues provided.
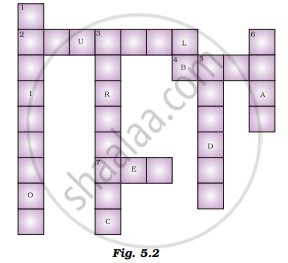
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour to the basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Curd contains ______ acid.
Which one of the following acids undergoes complete ionisation?
Exlain the following:
\[\ce{H3PO3}\] is not tribasic acid.
Draw the structure of Hydronium ion.
