Advertisements
Advertisements
प्रश्न
Classify the following reaction into different type:
CaO(s) + H2O(l) → Ca(OH)2(aq)
उत्तर
CaO(s) + H2O(l) → Ca(OH)2(aq)
This is an example of combination reaction.
APPEARS IN
संबंधित प्रश्न
Define a chemical reaction.
What do you observe when solid lead nitrate is heated?
What effect does an increase in concentration of H+ (aq.) in a solution have on the pH of solution?
Define: Chemical combination reaction
Calcium oxide reacts vigorously with water to produce slaked lime.
\[\ce{CaO{(s)} + H2O(l) -> Ca(OH)2(aq)}\]
This reaction can be classified as:
(A) Combination reaction
(B) Exothermic reaction
(C) Endothermic reaction
(D) Oxidation reaction
Classify the following reaction into –
- Direct combination
- Decomposition
- Displacement
- Double decomposition
The reaction is – Molten zinc at high temperatures, burns in air to give zinc oxide.
Give a balanced equation for –
A direct combination reaction involving two elements, one of which is a non-metal
Give a balanced equation for the following type of reaction:
A reaction of direct combination i.e. synthesis in which twp gases combine to give another gas – which turns lime water milky.
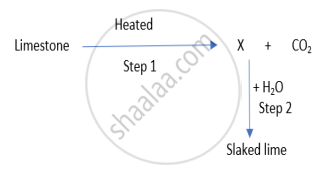
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
- Assertion: Rusting of Iron is endothermic in nature.
- Reason: As the reaction is slow, the release of heat is barely evident.
