Advertisements
Advertisements
प्रश्न
Compare the structures of H2O and H2O2.
संक्षेप में उत्तर
उत्तर
In gaseous phase, the water molecule has a bent form with a bond angle of 104.5°. The O–H bond length is 95.7 pm. The structure can be shown as:
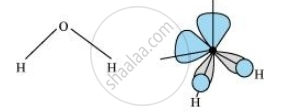
Hydrogen peroxide has a non-planar structure both in gas and solid phase. The dihedral angle in gas and solid phase is 111.5° and 90.2° respectively.
\[\begin{array}{cc}
\ce{H}\phantom{.......}\\
\backslash\phantom{......}\\
\ce{O - O}\\
\phantom{.......}\backslash\\
\phantom{........}\ce{H}
\end{array}\]
shaalaa.com
Water - Structure of Water
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
अध्याय 9: Hydrogen - EXERCISES [पृष्ठ २९८]
