Advertisements
Advertisements
प्रश्न
Complete the statement by filling in the blank with the correct word:
Decomposition of silver salts in the presence of sunlight is an example of _________.
विकल्प
double decomposition
photochemical decomposition
उत्तर
Decomposition of silver salts in the presence of sunlight is an example of photochemical decomposition.
APPEARS IN
संबंधित प्रश्न
What type of reaction is represented by the following equation?
NH4 CI → NH3 + HCI
What type of chemical reaction take place when electricity is passed through water?
Name the product formed on strongly heating ferrous sulphate crystals. What type of chemical reaction occurs in this change?
When a green iron salt is heated strongly, its colour finally changes to brown and odour of burning sulphur is given out.
(a) Name the iron salt.
(b) Name the type of reaction that takes place during the heating of iron salt.
(c) Write a chemical equation for the reaction involved.
Identify the type of following reaction :
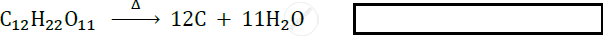
Give a balanced equation for the following type of reaction:
A thermal decomposition reaction in which a metallic nitrate decomposes to give – a basic oxide.
Differentiate between the following:
Electrolytic decomposition and photochemical decomposition.
The chemical reaction in which two or more products are formed from a single reactant is called _______ reaction.
Give the ratio in which hydrogen and oxygen are present in water by volume.
Marble statues are corroded or stained rain water. Identify the main reason.