Advertisements
Advertisements
प्रश्न
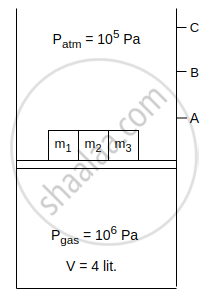
Consider an isothermal cylinder and massless piston assembly in which ideal gas is filled. Cross-sectional area of the cylinder = 1 m2. Three masses m1, m2 and m3 are kept on the piston. When m1 is removed, the piston moves upto point A. When m1 and m2 both are removed piston moves upto point B and when m1, m2 and m3 all three are removed, piston moves upto point C. The work done by the gas is ______ J, when piston moves from point B to point C.
[Given : m1 = 2 × 104 kg, m2 = 3 × 104 kg, g = 10 m/s2]
विकल्प
1000
1053
1500
3200
उत्तर
Consider an isothermal cylinder and massless piston assembly in which ideal gas is filled. Cross-sectional area of the cylinder = 1 m2. Three masses m1, m2 and m3 are kept on the piston. When m1 is removed, the piston moves upto point A. When m1 and m2 both are removed piston moves upto point B and when m1, m2 and m3 all three are removed, piston moves upto point C. The work done by the gas is 3200 J, when piston moves from point B to point C.
Explanation:
Initially piston is at equation,
`10^6 = ((2 xx 10^4 + 3 xx 1064 + "m"_3)xx10)/1 + 10^5`
`"m"^3 = ((10^6 - 10^5)/10) - 5 xx 10^4`
= 104 (10 - 1) - 5 × 104
= 104 (9 - 5) = 4 × 104 kg
since T = constant
PiVi = PBVB
`10^6 xx 4 = (10^5 + 4 xx 10^5) xx "V"_"B"`
`"V"_"B" = (4 xx 10^6)/(10^5 xx 5) = 40/5` = 8 lit.
Also,
PBVB = PCVC
104 × 5 × 8 = 105 × VC
VC = 40 L
WBC = - Pext (VC - VB)
= - 105 (40 - 8) × 10-3
= - 3200 J
Wby = 3200 J
