Advertisements
Advertisements
प्रश्न
Consider the figure and mark the correct option.
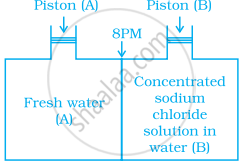
विकल्प
Water will move from side (A) to side (B) if a pressure lower than osmotic pressure is applied on piston (B).
Water will move from side (B) to side (A) if a pressure greater than osmotic, pressure is applied on piston (B).
Water will move from side (B) to side (A) if a pressure equal to osmotic pressure is applied on piston (B).
water will move from side (A) to side (B) if pressure equal to osmotic pressure is applied on piston (A).
उत्तर
Water will move from side (B) to side (A) if a pressure greater than osmotic, pressure is applied on piston (B).
Explanation:
Water will move from side 3 (concentrated sodium chloride solution) to side A (freshwater) if a pressure greater than osmotic pressure is applied on piston B.
APPEARS IN
संबंधित प्रश्न
Which of the two components of starch is water soluble?
An unripe mango placed in a concentrated salt solution to prepare pickle, shrivels because ______.
Water retention or puffiness due to high salt intake occurs due to ______.
Why are aquatic species more comfortable in cold water in comparison to warm water?
When kept in water, raisin swells in size. Name and explain the phenomenon involved with the help of a diagram. Give three applications of the phenomenon.
Evaporation of water is?
Molal enthalpy of fusion of water at 273 k is 6.0246 kg mol–1 calculated molal depression constant.
