Advertisements
Advertisements
प्रश्न
Define a combination reaction. Give one example of an exothermic combination reaction.
उत्तर
A chemical reaction in which 2 or more reactants combine to form a single product, the reaction is known as combination reaction. Most of the combination reactions are exothermic because they involve the formation of new bonds. For example,
\[\ce{H2_{(g)} + Cl2_{(g)} -> 2HCl_{(g)}}\]
\[\ce{SiO2_{(s)} + CaO_{(s)} -> CaSiO3_{(s)}}\]
APPEARS IN
संबंधित प्रश्न
Write the balanced chemical equation for the following and identify the type of reaction.
\[\ce{Hydrogen(g) + Chlorine(g) -> Hydrogen chloride (g)}\]
Find the odd man out:
Camphor, Ammonium Chloride, Naphthalene balls, Sugar
What type of chemical reaction take place when a magnesium wire is burnt in air?
What type of chemical reaction take place when ammonia and hydrogen chloride are mixed?
Classify the following reaction as combination, decomposition, displacement, precipitation and neutralization. Also balance the equation.
\[\ce{CaCO3_{(s)} ->[heat]CaO_{(s)} + CO2_{(g)}}\]
The Salt Story
From: The New Indian Express 9 March 2021
The salt pans in Marakkanam, a port town about 120 km from Chennai are the third largest producer of salt in Tamil Nadu. Separation of salt from water is a laborious process and the salt obtained is used as raw materials for manufacture of various sodium compounds.
One such compound is Sodium hydrogen carbonate, used in baking, as an antacid and in soda acid fire extinguishers.
The table shows the mass of various compounds obtained when 1litre of sea water is evaporated.
| COMPOUND | FORMULA | MASS OF SOLID PRESENT /g |
| Sodium Chloride | NaCl | 28.0 |
| Magnesium Chloride | MgCl2 | 8.0 |
| Magnesium Sulphate | MgSO4 | 6.0 |
| Calcium Sulphate | CaSO4 | 2.0 |
| Calcium Carbonate | CaCO3 | 1.0 |
| TOTAL AMOUNT OF SALT OBTAINED | 45.0 | |
Which compound in the table reacts with acids to release carbon dioxide?
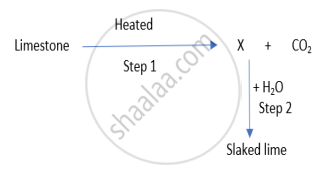
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
Read the text below and answer the questions that follow:
A small amount of hydrochloric acid was taken in a test tube. The test tube was heated. A glass rod was dipped in the ammonia solution and held on the top of the test tube. A white smoke was seen emanating from the tip of the glass rod.
- What must have happened?
- Which colour of gas is formed?
- Write the chemical equation for the reaction.
A clear solution of slaked lime is made by dissolving Ca(OH)2 in an excess of water. This solution is left exposed to air. The solution slowly goes milky as a faint white precipitate forms. Explain why a faint white precipitate forms, support your response with the help of a chemical equation.
A metal ribbon 'X' bums in oxygen with a dazzling white flame forming a white ash 'Y'. The correct description of X, Y and the type of reaction is:
