Advertisements
Advertisements
प्रश्न
Describe an experiment to prove the following:
HCI gas is heavier than air.
उत्तर
Two gas jars A and B are taken. A is filled with air and B with dry HCI gas. B is inverted over A, kept for few minutes and then jar is removed. Now a rod dipped in ammonium hydroxide is introduced into the jar A, white fumes are seen. This shows that HCI displaces the air since it is heavier than air.
APPEARS IN
संबंधित प्रश्न
Study the figure given below and answer the questions which follow:
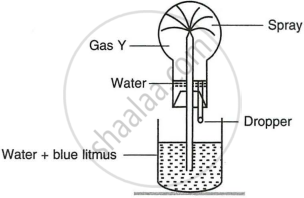
- Identify the gas Y.
- What property of gas Y does this experiment demonstrate?
- Name another gas which has the same property and can be demonstrated through this experiment.
Name the drying agent used to dry HCl gas.
How is the gas collected and why?
How can you prove that hydrochloric acid contain :
Chlorine?
How will you show that hychloric acid contains both hydrogen and chlorine (other than by electrolysis) ?

The diagram shows an apparatus for the laboratory preparation of hydrogen chloride.
(i) Identify A and B
(ii) Write the equation for the reaction
(iii) How would you check whether or not the gas jar is filled with hydrogen chloride?
(iv) What does the method of collection tell you about the density of hydrogen chloride.
The following question is pertaining to the laboratory preparation of hydrogen chloride gas.
Write the equation for its preparation mentioning the condition required.
What property of hydrogen chloride is demonstrated when it is collected by downward delivery (Upward displacement)?
State the following:
The drying agent used in the laboratory preparation of HCl gas.
Identify the terms for the following:
A method used to collect HCl gas.
