Advertisements
Advertisements
प्रश्न
Describe Gabriel's phthalimide synthesis. (Give reaction)
उत्तर
A nucleophile in the form of an imide ion has formed the reaction of KOH with phthalimide. The nucleophilic imide ion attacks the electrophilic carbon of the alkyl halide. The nitrogen atom subsequently replaces the halogen (Fluorine, Chlorine, Bromine or Iodine) in the alkyl halide and bonds with the carbon itself. This results in the formation of an N-Alkyl Phthalimide.
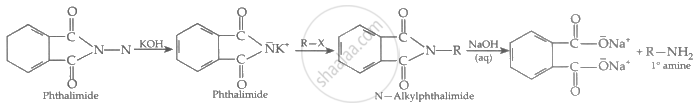
Hydrolysis of this phthalimide yields a primary alkyl amine. In other words, in the process of hydrolysis of phthalimide, the hydroxide ion attacks the carbon atom bonded to the nitrogen atom, cleaving the N-Alkyl phthalimide and the cation in the base attaches itself to the oxygen atom as well. However, Aryl amines cannot be prepared via Gabriel synthesis as aryl halides do not undergo simple nucleophilic substitution.
