Advertisements
Advertisements
प्रश्न
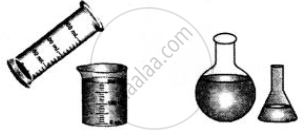
Describe simple experiments to prove that liquids have no definite shape
उत्तर
have no definite shape
Experiment: A liquid poured into any container takes up the shape of each container.
Conclusion: All liquids have no definite shape.
संबंधित प्रश्न
Give one difference between atoms and molecules.
Fill in the blanks:
Any matter which has a definite ______ but no definite shape is called a ______.
Write whether the following statement is true or false:
If the container in which a gas is collected has an opening, the gas will flow out and spread itself indefinitely
Describe simple experiments to prove that solids have a definite volume
In solids, the space between the particles is less than in ______.
Match the following.
| Srl. no. | A | B | C |
| i | Separation of visible undesirable components | Water mixed with chalk powder | Magnetic Separation |
| ii | Separation of heavier and lighter components | Sand and water | Decantation |
| iii | Separation of insoluble impurities | Iron impurities | Filtration |
| iv | Separation of magnetic components from non-magnetic components | Rice and stone | Hand-picking |
| v | Separation of solids from liquids | Husk and paddy | Winnowing |
The feather and paper cup can float on the water.
What is the physical state of water at 100ºC?
A strong current of trivalent gaseous boron passed through a silicon crystal decreases the density of the crystal due to part replacement of silicon by boron and due to interstitial vacancies created by missing Si atoms. In one such experiment, one gram of silicon is taken and the boron atoms are found to be 1000 ppm by weight, when the density of the Si crystal decreases by 12%. The percentage of missing vacancies due to silicon, which are filled up by boron atoms will be ____ g. [Given · Atomic wt. Si= 30, B = 11]
The density of solid Argon is 1.6 ml at - 233°C. If the Argon atom is assumed to be sphere of radius 1.5 × 10-8 cm, then the ______ % of solid Argon is apparently occupied.
[Take : NA = 6 × 1023, Atomic mass of Ar = 40]
