Advertisements
Advertisements
प्रश्न
Describe the Brownian motion.
उत्तर
Brownian motion is due to the bombardment of Brownian motion suspended particles by molecules of the surrounding fluid. But during the 19th century, people did not accept that every matter is made up of small atoms or molecules. In the year 1905, Einstein gave the systematic theory of Brownian motion based on kinetic theory and he deduced the average size of molecules.
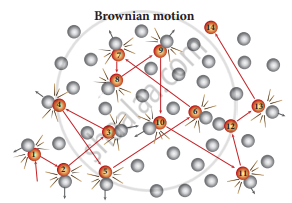
Particles in Brownian motion
According to kinetic theory, any particle suspended in a liquid or gas is continuously bombarded from all directions so that the mean free path is almost negligible. This leads to the motion of the particles in a random and zig-zag manner. But when we put our hand in the water it causes no random motion because the mass of our hand is so large that the momentum transferred by the molecular collision is not enough to move our hand.
Factors affecting Brownian Motion:
- Brownian motion increases with increasing temperature.
- Brownian motion decreases with bigger particle size, high viscosity and density of the liquid (or) gas.
