Advertisements
Advertisements
प्रश्न
Differentiate between: Gas and vapour
उत्तर
| Gas | Vapourisation |
| The substance which remain in the gaseous state under normal conditions of temperature and pressure are called gases | The process by which a substance changes from a liquid state to vapour state is called vaporisation or evaporation |
| example Oxygen, hydrogen, nitrogen | example water to steam |
APPEARS IN
संबंधित प्रश्न
What do you understand by the change of phase of a substance?
What should be added to a Celsius scale reading so as to obtain the corresponding Kelvin scale reading ?
State one condition necessary to liquefy gases (other than applying high pressure).
Fill in the following blank with suitable words :
Scientists say that there are actually five states of matter : solid, liquid, gas, .................... and ...................
Define 'boiling point' of a substance ? What is the boiling point of water ?
Give reasons:
Why a solid does not flow, but a liquid flows?
What will happen if....? And why?
A bell is made of wood.
Why does this happen?
What you see in the picture.
State which of the three states of matter Le. solids, liquids or gases – have
(a) No definite volume
(b) A definite shape
(c) High density
(d) No free surfaces
(e) Particles – which diffuse very easily.
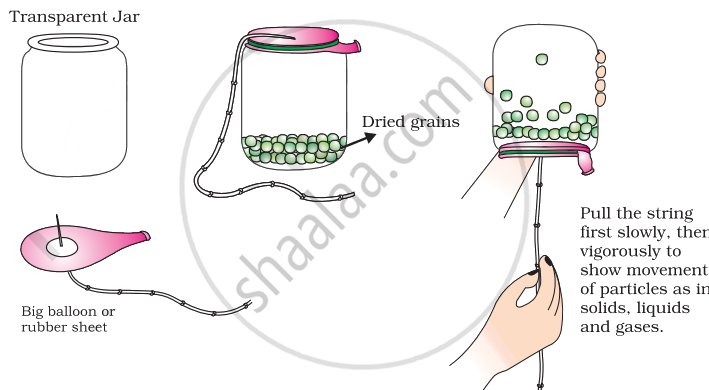
Prepare a model to demonstrate movement of particles in solids, liquids and gases.
For making this model you will need
- A transparent jar
- A big rubber balloon or piece of stretchable rubber sheet
- A string
- Few chickpeas or black gram or dry green peas.
How to make?
- Put the seeds in the jar.
- Sew the string to the centre of the rubber sheet and put some tape to keep it tied securely.
- Stretch and tie the rubber sheet on the mouth of the jar.
- Your model is ready. Now run your fingers up and down the string by first tugging at it slowly and then rapidly.