Advertisements
Advertisements
प्रश्न
Discuss the spectral series of hydrogen atom.
उत्तर
- As electron in excited states have a very small lifetime, these electrons jump back to the ground state through spontaneous emission in a short duration of time (approximately 10) by emitting the radiation with the same wavelength (or frequency) corresponding to the colours is absorbed. This is called emission spectroscopy.
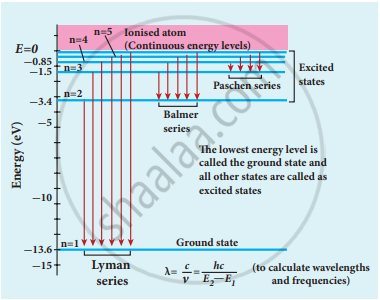

Spectral series – Lyman, Balmer, Paschen series - In each series, the distance of separation between the consecutive wavelengths decreases from higher wavelength to the lower wavelength, and also wavelength in each series approach a limiting value known as the series limit.
- The wavelengths of these spectral lines perfectly agree with the wavelengths calculate using an equation derived from the Bohr atom model.
`1/lambda = "R" (1/"n"^2 - 1/"m"^2) = bar"v"` ...(1)
where `bar"v"` is known as wave number which is inverse of the wavelength, R is known as Rydberg constant whose value is 1.09737 × 107 m-1 and m and n are positive integers such that m > n.
The various spectral series are discussed below:
- Lyman series:
Put n = 1 and m = 2, 3, 4 …..in equation (1). The wave number or wavelength of spectral lines of Lyman series which lies in the ultra-violet region is
`bar"v" 1/lambda (1/"n"^2 - 1/"m"^2) = bar"v"` - Balmer series:
Put n = 2 and m = 3, 4, 5 …. in equation (1). The wave number or wavelength of spectral lines of Balmer series which lies in the visible region is
`bar"v" 1/lambda "R"(2/"n"^2 - 1/"m"^2) = bar"v"` - Paschen series:
Put n = 3 and m = 4, 5, 6…. in equation (1). The wave number or wavelength of spectral lines of Paschen series which lies in the infra-red region (near IR) is
`bar"v" 1/lambda "R"(3/"n"^2 - 1/"m"^3) = bar"v"` - Brackett series:
Put n = 4 and m = 5, 6, 7 ….in equation (1). The wave number or wavelength of spectral lines of Brackett series which lies in the infra-red region (middle IR) is
`bar"v" 1/lambda "R"(4/"n"^2 - 1/"m"^3) = bar"v"` - Pfund series:
Put n = 5 and m = 6, 7, 8 …. in equation (1). The wave number or wavelength of spectral lines of Pfund series which lies in the infra-red region (far IR) is
`bar"v" 1/lambda "R"(5/"n"^2 - 1/"m"^2) = bar"v"`
- Lyman series:
APPEARS IN
संबंधित प्रश्न
According to the Bohr Theory, which of the following transitions in the hydrogen atom will give rise to the least energetic photon?
In J.J. Thomson e/m experiment, a beam of electron is replaced by that of muons (particle with same charge as that of electrons but mass 208 times that of electrons). No deflection condition is achieved only if
Give the results of Rutherford alpha scattering experiment.
Write down the postulates of Bohr atom model.
What is meant by excitation energy?
What is distance of closest approach?
Explain the J. J. Thomson experiment to determine the specific charge of an electron.
Derive the energy expression for an eletron is the hydrogen atom using Bohr atom model.
In the Bohr atom model, the frequency of transitions is given by the following expression
v = `"Rc" (1/"n"^2 - 1/"m"^2)`, where n < m
Consider the following transitions:
| Transitions | m → n |
| 1 | 3 → 2 |
| 2 | 2 → 1 |
| 3 | 3 → 1 |
Show that the frequency of these transitions obey sum rule (which is known as Ritz combination principle).
- A hydrogen atom is excited by radiation of wavelength 97.5 nm. Find the principal quantum number of the excited state.
- Show that the total number of lines in emission spectrum is `("n"("n - 1"))/2`.
Compute the total number of possible lines in emission spectrum as given in(a).
