Advertisements
Advertisements
प्रश्न
Does the substance absorb or liberate any heat energy during the change of phase?
उत्तर
Yes, the substance absorbs or liberates heat during the change of phase.
APPEARS IN
संबंधित प्रश्न
Convert the following temperature to Celsius scale:
573 K
Fill in the following blank with suitable words :
The state of matter called ........................ makes a fluorescent tube (or neon sign bulb) to glow.
Explain why, ice at 0° C is more effective in cooling than water at the same temperature.
Draw a labelled diagram of the experimental set-up to study the latent heat of vaporisation of water.
Which of the following factors are responsible for the change in state of solid carbon dioxide when kept exposed to air ?
- increase in pressure
- ncrease in temperature
- decrease in pressure
- decrease in temperature
Give reasons:
How is interconversion of states of matter different from chemical reaction?
Name the phenomenon which causes the following changes:
Conversion of water into steam
A kind of matter which can sublime is
Explain the term vaporization
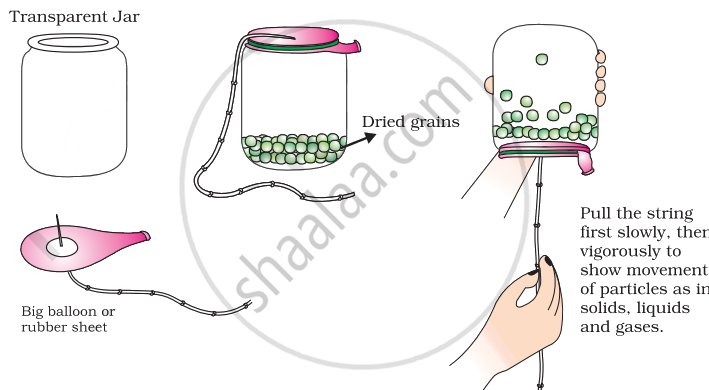
Prepare a model to demonstrate movement of particles in solids, liquids and gases.
For making this model you will need
- A transparent jar
- A big rubber balloon or piece of stretchable rubber sheet
- A string
- Few chickpeas or black gram or dry green peas.
How to make?
- Put the seeds in the jar.
- Sew the string to the centre of the rubber sheet and put some tape to keep it tied securely.
- Stretch and tie the rubber sheet on the mouth of the jar.
- Your model is ready. Now run your fingers up and down the string by first tugging at it slowly and then rapidly.