Advertisements
Advertisements
प्रश्न
Draw structures 01 conjugate bases of monochloroacetic acid and dichloroacetic acid. Which one is more stabilized by -I effect?
संक्षेप में उत्तर
उत्तर
- \[\ce{\underset{Acid}{CiC}H2 - COOH + H2O <=> \underset{Conjugate base}{ClCH2 –COO ^–} + H3O^+}\]
ClCH2COO – is a conjugate base of ClCH2COOH - \[\ce{Cl2CH - COOH + H2O <=> \underset{Conjugate base}{ClCHCOO –} + H3O^+}\]
ClCHCOO – is a conjugate base of Cl2CHCOOH 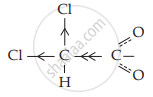
The dichloroacetate ion formed gets stabilised due to electron-withdrawing effect of two chlorine atoms ( – I effect).
shaalaa.com
Chemical Properties of Carboxylic Acids
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
