Advertisements
Advertisements
प्रश्न
Draw the chain isomers of hexane \[\ce{(C6H14)}\].
उत्तर १
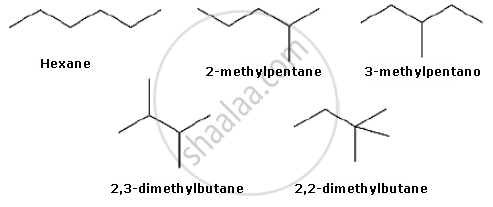
उत्तर २
- \[\ce{\underset{n-hexane}{H3C - H2C - H2C - H2C - H2C - CH3}}\]
- \[\begin{array}{cc}
\ce{H3C - HC - H2C - H2C - CH3}\\
|\phantom{................}\\
\ce{\underset{iso-hexane}{H3C}\phantom{..............}}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H3C}\phantom{.....}\\
|\phantom{.......}\\
\ce{H3C - C - H2C - CH3}\\
|\phantom{.......}\\
\ce{H3C}\phantom{.....}\\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Draw structural formula of the compound isomer of n-butane ?
What is used to describe these compounds taken together?
Why was vital force theory discarded ?
Define or explain chain isomerism with an example.
What is hydrogenation? How hydrogenation of ethene occurs? Give one use of hydrogenation.
Fill in the blank with appropriate word/words.
Two isomers of the compound having molecular formula C3H6O are __________and ________
For each of the compounds : (a) ethane (b) vinegar (c) Marsh gas draw the relevant structural formula
The list of some organic compounds is given below:
Ethanol, ethane, methane, ethyne and ethene
From the above list, name a compound:
Formed by the dehydration of ethanol by concentrated sulphuric acid.
The number of C-H bonds in ethane molecule are ______.
Name the following:
A hydrocarbon which contributes towards the greenhouse effect
Name the compound prepared by the following reaction:
C2H5Br + KOH (alcoholic solution) →
