Advertisements
Advertisements
प्रश्न
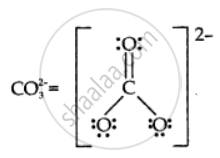
Draw the Lewis structures for the given molecule and ion:
उत्तर
APPEARS IN
संबंधित प्रश्न
Write Lewis dot symbols for atoms of the following elements: Mg, Na, B, O, N, Br.
Draw the Lewis structures for the following molecule and ion:
H2S
Draw the Lewis structures for the given molecule and ion:
SiCl4
Draw the Lewis structures for the given molecule and ion:
BeF2
Draw the Lewis structures for the following molecule and ion:
HCOOH
Explain why BeH2 molecule has a zero dipole moment although the Be–H bonds are polar.
According to Lewis theory, a base is any species which ____________.
Write Lewis structure of the following compounds and show formal charge on atom.
Write Lewis structure of the following compounds and show formal charge on atom.
Write Lewis structure of the following compounds and show formal charge on atom.
Represent diagrammatically the bond moments and the resultant dipole moment in
Which of the following species contains equal number of σ– and π–bonds?
