Advertisements
Advertisements
प्रश्न
Excess of isobutane on reactions with Br2 in presence of light at 125°C gives which one of the following, as the major product?
विकल्प
\[\begin{array}{cc}\phantom{....}\ce{CH3}\\
\phantom{..}|\\\ce{CH3 - C - Br}\\ \phantom{..}|\\\phantom{....}\ce{CH3}\end{array}\]\[\begin{array}{cc}\ce{CH3 - CH - CH2 - Br}\\
|\phantom{.......}\\\ce{CH3}\phantom{.....}\end{array}\]\[\begin{array}{cc}\ce{CH3 - CH - CH2 - Br}\\
|\phantom{.......}\\\phantom{.}\ce{CH2 - Br}\end{array}\]\[\begin{array}{cc}\ce{Br}\phantom{....}\\
|\phantom{.....}\\\ce{CH3 - C - CH2 - Br}\\
|\phantom{.....}\\\ce{CH3}\phantom{..}\end{array}\]
उत्तर
\[\begin{array}{cc}\phantom{....}\ce{CH3}\\
\phantom{..}|\\\ce{CH3 - C - Br}\\ \phantom{..}|\\\phantom{....}\ce{CH3}\end{array}\]
Explanation:
We can consume excessive amounts of alkane, which results in the formation of monobromo product, in order to avoid polybromo product.
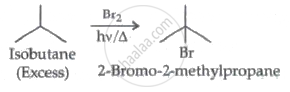
It is a free radical mechanism in which Br can attack on the available sites.
