Advertisements
Advertisements
प्रश्न
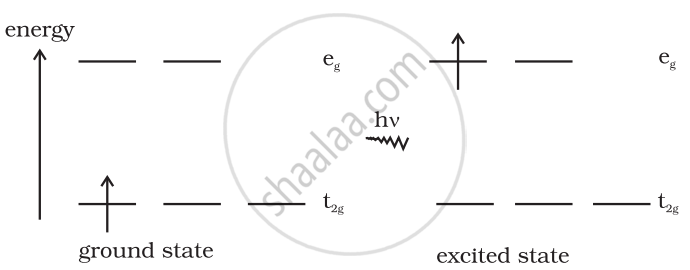
Explain the violet colour of the complex [Ti(H2O)6]3+ on the basis of crystal field theory.
उत्तर
Coordination compounds exhibit colours attributed to the crystal field theory, which correspond to the d-d transitions of the elements. For example, excitation of an electron from vacant state e.g. absorbing light will excite the electron from t2g level. Now, in case of [Ti(H2O)6]3+ when light corresponding to the energy of the yellow-green region is absorbed by the complex, it will excite the electron from t2g level to a level. Thus, the complex will be violet in colour.
संबंधित प्रश्न
Which of the following complexes will give a white precipitate on treatment with a solution of barium nitrate?
- [Cr(NH3)4SO4] Cl
- [Co(NH3)4Cl2] NO2
- [Cr(NH3)4Cl2]SO4
- [CrCl2(H2O)4]Cl
On adding NaOH to ammonium sulphate, a colourless gas with pungent odour is evolved, which forms a blue-coloured complex with Cu2+ ion. Identify the gas.
A solution of [Ni(H2O)6]2+ is green but a solution of [Ni(CN)4]2− is colourless. Explain.
[Fe(CN)6]4− and [Fe(H2O)6]2+ are of different colours in dilute solutions. Why?
What will be the correct order for the wavelengths of absorption in the visible region for the following?
[Ni(NO2)6]4−, [Ni(NH3)6]2+, [Ni(H2O)6]2+
Why a solution of [Ni(H2O)6]2+ is green while a solution of [Ni(CN)4]2− is colourless? (At. no. of Ni = 28)
Which of the following is the most likely structure of CrCl3.6H2O if `1/3` of total chlorine of the compound is precipitated by adding AgNO3 to its aqueous solution?
The wavelength of light absorbed by [Cu(H2O)4]2+ is 600 nm. The crystal field splitting energy is ______
A solution of [Ni(H2O)6]2+ is green, but a solution of [Ni(CO)4] is colourless. Explain.
[Atomic number: Ni = 28]
Aqueous copper sulphate solution (blue in colour) gives a bright green solution with aqueous potassium chloride. Explain this experimental result.
