Advertisements
Advertisements
प्रश्न
Explain various types of constitutional isomerism (structural isomerism) in organic compounds.
उत्तर
Structural isomerism:
This type of isomers have the same molecular formula but differs in their bonding sequence.
- Chain or nuclear or skeletal isomerism:
These isomers differ in the way in which the carbon atoms are bonded to each other in a carbon chain or in other words isomers have similar molecular formula but differ in the nature of the carbon skeleton (ie. Straight or branched).
\[\ce{CH3 - CH2 - CH2 - CH2 - CH3}\]
n - Pentane
\[\begin{array}{cc}\ce{CH3 - CH - CH2 - CH3}\\|\phantom{.........}\\\ce{CH3}\phantom{......}\end{array}\]
iso pentane (or) 2 - methyl butane
\[\begin{array}{cc}\phantom{...}\ce{CH3}\\|\\\ce{CH3 - C - CH3}\\|\\\phantom{...}\ce{CH3}\end{array}\]
neopentane (or) 2, 2 - dimethyl propane - Position isomerism:
If different compounds belonging to same homologous series with the same molecular formula and carbon skeleton, but differ in the position of substituent or functional group or an unsaturated linkage are said to exhibit position isomerism.
Molecular formula C5H10
\[\ce{CH3 - CH2 \underset{\text{pent-l-ene}}{- CH2 -} CH = CH2}\] and
\[\ce{CH3 - CH2 \underset{\text{pent-l-ene}}{- CH = C}H - CH3}\] - Functional isomerism:
Different compounds having same molecular formula but different functional groups are said to exhibit functional isomerism.
Molecular formula C3H6O
CH3 – CH2 – CHO
propanal (aldehyde group)
\[\begin{array}{cc}\ce{O}\\||\\\ce{\underset{\text{propanone (keto group)}}{CH3 - C - CH3}}\end{array}\]
APPEARS IN
संबंधित प्रश्न
Which one of the following shows functional isomerism?
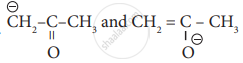 are
are
Nitrogen detection in an organic compound is carried out by Lassaigne’s test. The blue colour formed is due to the formation of
An organic Compound weighing 0.15g gave on carius estimation, 0.12g of silver bromide. The percentage of bromine in the Compound will be close to ______.
Identify the functional group in the following compound.
Acetaldehyde
Identify the functional group in the following compound.
Oxalic acid
Identify the functional group in the following compound.
Di methyl ether
Give the general formula for the following class of organic compound.
Aliphatic monohydric alcohol
Give the general formula for the following class of organic compound.
Aliphatic ketones
Describe optical isomerism with a suitable example.
