Advertisements
Advertisements
प्रश्न
Explain, why the following statement is not correct:
Lead chloride can be prepared by adding dilute HCI to lead sulphate solution.
उत्तर
Lead chloride cannot be prepared by adding dilute HCI to lead sulphate solution because lead sulphate is insoluble salt. Infact it can be prepared by adding dilute HCI to lead nitrate solution since lead nitrate is soluble compound.
APPEARS IN
संबंधित प्रश्न
Certain blank spaces are left in the following table and these are labelled as A, B, C, D
and E. Identify each of them
| Lab preparation of | Reactants used | Products formed | Drying Agent | Method of collection |
|
| 1 | HCl gas | NaCl + H2SO4 |  |
conc. H2SO4 | 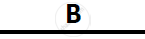 |
| 2 | NH3 gas |
|
Mg(OH)2 NH3 |
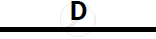 |
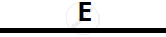 |
Identify the gas evolved and give the chemical test in the following cases
Dilute hydrochloric acid reacts with sodium sulphite.
State your observation in given case When dilute hydrochloric acid is added to sodium carbonate crystals
Give the balanced equation for the laboratory preparation of hydrogen chloride gas reaction.
Explain why a solution of hydrogen chloride in water turns blue litmus red and conducts electricity, while a solution of the same gas in toluene:
- has no effect on litmus, and
- does not conduct electricity.
The drying agent used to dry \[\ce{HCl}\] gas is ______.
Draw a labelled diagram and explain the laboratory preparation of hydrogen chioride gas.
Explain, why (or give reasons for)
In the preparation chloride from sodium chloride, the gas can be obtained below 200°C or above. But the lower temperature is preferred.
Choose the correct answer from the options given below:
Bleaching powder reacts with few drops of concentrated HCl to give
Complete and balance the equations:
KMnO4 + HCI → _______+ _______ + _______ + ______ + 8H2O
Write the equation for the reaction which takes place in question(a).
One chemical test that would enable you to distinguish between the following pair of chemicals. Describe what happens with each chemical or state 'no visible reaction'.
Sodium chloride solution and sodium nitrate solution.
One chemical test that would enable you to distinguish between the following pair of chemicals. Describe what happens with each chemical or state 'no visible reaction'.
Sodium chloride solution and sodium nitrate solution.
Give one test to distinguish between the following pair of chemicals.
Sodium nitrate solution and sodium chloride solution.
Give one test to distinguish between the following pair of chemicals.
Iron (III) Chloride solution and copper chloride solution.
Answer the following question related to the laboratory preparation of the hydrogen chloride gas:
State the temperature required in the preparation.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
