Advertisements
Advertisements
प्रश्न
Explain why Grignard reagents should be prepared under anhydrous conditions?
उत्तर
Grignard reagents are very reactive. In the presence of moisture, they react to give alkanes.
\[\ce{\underset{reagent}{\underset{Grigrard}{\overset{\delta-}{R} \overset{\delta +}{M}g \overset{\delta -}{X}}} + H2O -> \underset{Alkane}{R - H} + Mg (OH) X}\]
Therefore, Grignard reagents should be prepared under anhydrous conditions.
APPEARS IN
संबंधित प्रश्न
Identify A, B, C, D, E, R and R1 in the following:

How will you bring about the following conversion?
Bromomethane to propanone
How will you bring about the following conversion?
1-Chlorobutane to n-octane
What happens when methyl bromide is treated with sodium in the presence of dry ether?
Write the structure of main compounds A and B in the following reaction: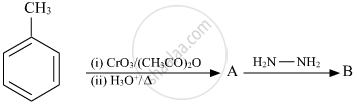
A Grignard reagent may be made by reacting magnesium with ____________.
Why is it necessary to avoid even traces of moisture during the use of a Grignard reagent?
Which of the statements about Grignard reagent is false?
\[\ce{R - CH2 - CCl2 - R ->[Peagent] R - C ≅ C - R}\]. The reagent is:-
Grignard reagent shows addition on
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
Explain why Grignard reagents should be prepared under anhydrous conditions?
