Advertisements
Advertisements
प्रश्न
Explain why the following system is not aromatic?
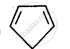
संक्षेप में उत्तर
उत्तर

For the given compound, the number of π-electrons is four.
By Huckel’s rule,
4n + 2 = 4
4n = 2
n = 1/2
For a compound to be aromatic, the value of n must be an integer (n = 0, 1, 2…), which is not true for the given compound. Hence, it is not aromatic in nature.
shaalaa.com
Aromatic Hydrocarbons - Aromaticity (Huckel Rule)
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
अध्याय 13: Hydrocarbons - EXERCISES [पृष्ठ ४०५]
APPEARS IN
संबंधित प्रश्न
What are the necessary conditions for any system to be aromatic?
Explain why the following system is not aromatic?

Explain why the following system is not aromatic?
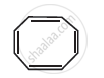
Four structures are given in options (i) to (iv). Examine them and select the aromatic structures.
| (i) | 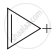 |
| (ii) | 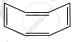 |
| (iii) | 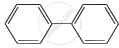 |
| (iv) |  |
Cyclopentadienyl anion is ______.
Among the following, the aromatic compounds are:
| (A) | 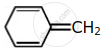 |
| (B) |  |
| (C) | 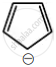 |
| (D) | 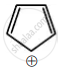 |
Choose the correct answer from the following options:
