Advertisements
Advertisements
प्रश्न
Fill in the blank.
As we descend the electrochemical series containing cations, the tendency of the cations to get ________ at the cathode increases.
विकल्प
oxidized
reduced
उत्तर
As we descend the electrochemical series containing cations, the tendency of the cations to get reduced at the cathode increases.
APPEARS IN
संबंधित प्रश्न
Electrolysis of aqueous sodium chloride solution will form ________ at the cathode. (Hydrogen gas / Sodium metal)
Name a salt which is a weak electrolyte.
Explain, why solid sodium chloride does not allow electricity to pass through?
Aqueous solution of nickel sulphate contains \[\ce{Ni^{+2}}\] and \[\ce{SO^{-2}_{4}}\] ions.
- Which ion moves towards the cathode?
- What is the product at the anode?
Differentiate between the electrical conductivity of copper sulphate solution and copper metal.
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
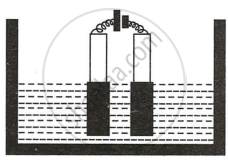
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
M is a metal above hydrogen in the activity series and its oxide has the formula M2O. The oxide when dissolved in water forms the corresponding hydroxide which is a good conductor of electricity. In the above context answer the following:
- What kind of combination exists between M and O?
- How many electrons are there in the outermost shell of M?
- Name the group to which M belongs.
- State the reaction taking place at the cathode.
- Name the product at the anode.
Give appropriate scientific reasons for the following statement:
During electrolysis of molten lead bromide, graphite anode is preferred to other electrodes.
Copper sulphate solution is electrolysed using copper electrodes.
Which electrode [cathode or anode] is the oxidizing electrode? Why?
Classify the following substance:
Dilute sulphuric acid
