Advertisements
Advertisements
प्रश्न
Fill in the following blank using suitable word:
Whenever mechanical work is done, .......... Is produced.
उत्तर
Whenever mechanical work is done, heat Is produced.
APPEARS IN
संबंधित प्रश्न
200 g of hot water at 80°C is added to 400 g of cold water at 10°C. Neglecting the heat taken by the container, calculate the final temperature of the mixture of water. Specific heat capacity of water = 4200 J kg-1K-1.
Describe an experiment to show that there is absorption of heat energy when the ice melts.
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
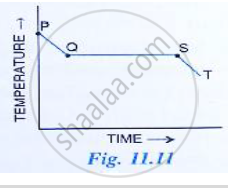
The melting point of naphthalene, a crystalline solid is 80℃ and the room temperature is 30℃. A sample of liquid naphthalene at 100℃ is cooled down to the room temperature. Draw a temperature time graph to represent this cooling.
(i) Define Calorimetry.
(ii) Name the material used for making a Calorimeter.
(iii) Why is a Calorimeter made up of thin sheets of the above material answered in (ii).
Give scientific reasons for the following:
It is much easier to skate on rough ice than on glass.
1 kg of molten lead at its melting point of 327°C is dropped into 1 kg of water at 20°C. Assuming no loss of heat, calculate the final temp. of water. (Sp. heat of lead = 130 J/kg°C, latent heat of lead = 27000 J/kg and Sp. heat of water = 4200 J/kg°C).
Out of the three metals A, B and C of specific heat 900 J kg-1 °C-1, 380 J kg-1 °C-1 and 460 J kg-1 °C-1 respectively, which will you prefer for calorimeter? Given reason.
Why does the heat supplied to a substance during its change of state not cause any rise in its temperature?
