Advertisements
Advertisements
प्रश्न
Give a balanced equation for the following conversion:
Calcium sulphate in permanent hard water to calcium carbonate using sodium carbonate.
उत्तर
\[\ce{\underset{\text{Sodium carbonate}}{Na2CO3} + \underset{\text{In hard water}}{CaSO4} -> \underset{\text{Cal. carbonate}}{CaCO3↓} + \underset{\text{Sodium sulphate}}{Na2SO4}}\]
APPEARS IN
संबंधित प्रश्न
Match the statements in column A with that in column B.
| Column A | Column B |
| (a) deliquescent substance | (1) sodium bicarbonate |
| (b) hygroscopic substance | (2) magnesium chloride |
| (c) efflorestance substance | (3) conc. sulphuric acid |
| (d) substance causing temporary hardness | (4) washing soda |
Give one word for the following statement:
The gaseous form of water found in air .
The ultimate source of all water on the earth is
Explain why:
Water is an excellent liquid to use in cooling systems.
Explain any two environmental impacts of an ‘oil spill’.
State the importance of water for all general uses.
Give a reason for the following:
Efflorescence is minimum during humid conditions.
37% of our brain‛s mass consists of water.
The jumbled word is given here. Write the correct form of each word.
CHARREGE
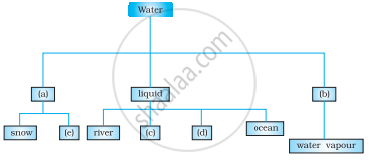
Complete the following chart by writing appropriate words in the boxes marked (a) to (e).