Advertisements
Advertisements
प्रश्न
Give a balanced equation for the following conversion: [In one or two steps]
A metallic oxide to calcium carbonate.
उत्तर
A metallic oxide to calcium carbonate
Step I: \[\ce{\underset{\text{Cal.oxide}}{CaO} + H2O -> Ca[OH]2}\]
Step II: \[\ce{Ca[OH]2 + CO2 ->\underset{\text{[Cal. carbonate]}}{CaCO3} + H2O}\]
APPEARS IN
संबंधित प्रश्न
Choose the correct alternative
The process by which decayed plants slowly convert into coal is called.
Explain the term ‘green house effect’. How can it be both beneficial and harmful for life on earth ?
FILL IN THE BLANK
Organic compounds have ........ boiling and melting points.
Write the name and the chemical formula of the simplest alkene.
Name of the following compounds :
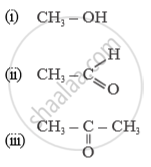
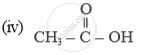
In which compound forms does carbon occur?
Select the appropeiate option of the following statement:
A nitrogenous fertilizer obtained from carbon dioxide.
Complete the statement by filling the blank with the correct word from the option.
Adsorption capacity of wood charcoal is increased by passing ______ over wood charcoal, at high temperature.
Carbon is one of the most important ______ elements.
The carbon cycle is the ______ cycle.
