Advertisements
Advertisements
प्रश्न
Give three differences between elements & compounds with relevant examples.
उत्तर
Three differences between elements and compounds :
Element :
- They contain one kind of atoms only e.g. element (Na) Sodium and [Cl] chlorine.
- The elements have their own set of properties e.g. [H] Hydrogen is combustible.
- They cannot be broken down into two or simpler substances by physical or chemical means.
Compound :
- They contain two or more kinds of atoms, e.g. compound — water [H2O].
- The Properties of compounds are entirely new i.e. water [H20] is a liquid and can extinguish the fire.
- It cannot be broken down into two or simpler substances by chemical means.
APPEARS IN
संबंधित प्रश्न
Find the atomicity of the Following molecule:
Calcium Chloride
Tick (√) the correct alternative from the choice given for the following statement.
The most abundant elements in the universe are
Tick (√) the correct alternative from the choice given for the following statement
The most ductile metal.
Complete the statement with the correct words.
__________ are chemicals formed on reaction of a base with an _____ giving salt and water.
Explain the difference between Metals and non-metals.
Explain the difference between Atoms and molecules.
Select the correct answer from the choice in the bracket.
The least reactive metal.
Select the correct answer from A, B, C, D & E for the statement given below :
A metalloid.
Select the correct answer from the choice given below to complete the sentence :
Dust in air is an example of _________ mixture.
Higher Order Thinking Skills.
In the diagram given below, the circle, square and triangle represent the atoms of different elements.
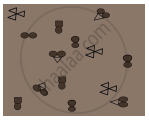
Identify all combinations that represent
- molecule of a compound
- molecule of an element consisting of two atoms
- molecule of an element consisting of three atoms
