Advertisements
Advertisements
प्रश्न
How does N/Z ratio affect nuclear stability? Explain with a suitable diagram.
उत्तर
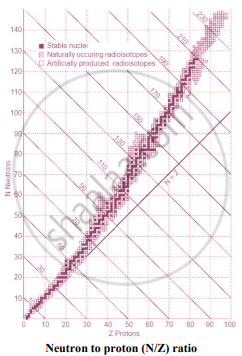
- When the graph of number of neutrons (N) against protons (Z) is drawn, and all the stable isotopes are plotted on it, there is quite a clear correlation between N and Z. This graph is shown in the adjacent figure.
- A large number of elements have several stable isotopes and hence, the curve appears as a belt or zone called stability zone. All stable nuclides fall with this zone and the nuclei that are to the left or to the right of the stability zone are unstable and exhibit radioactivity. Below the belt, a straight line which represents the ratio N/Z to be nearly unity (i.e., N = Z) is shown.
- For nuclei lighter than \[\ce{_20^40 Ca}\], the straight line (N = Z) passes through the belt. The lighter nuclides are therefore stable (N/Z being 1).
- The N/Z ratio for the stable nuclides heavier than calcium gives a curved appearance to the belt with a gradual increase of N/Z (> 1). The heavier nuclides, therefore, need more neutrons than protons to attain stability. The heavier nuclides with an increasing number of protons render large coulombic repulsions. With an increased number of neutrons, the protons within the nuclei get more separated, which renders them stable.
- Thus, nuclear stability is linked to the number of nucleons (neutrons and protons). In general, the lighter stable nuclei have equal numbers of protons and neutrons while heavier stable nuclei have increasingly more neutrons than protons.
APPEARS IN
संबंधित प्रश्न
On the basis of even-odd of protons and neutrons, what type of nuclides is most stable?
The nuclides with odd number of both protons and neutrons are the least stable. Why?
Referring the stability belt of stable nuclides, which nuclides are β– and β+ emitters? Why?
Explain with an example each nuclear transmutation and artificial radioactivity. What is the difference between them?
What is the binding energy per nucleon?
Explain with the help of a diagram how binding energy per nucleon affects nuclear stability?
Write two factors responsible for its stability.
The total binding energy of 58Ni is 508 MeV. What is its binding energy per nucleon?
A produces B by α-emission. If B is in group 16 of the periodic table, what is the group of A?
