Advertisements
Advertisements
प्रश्न
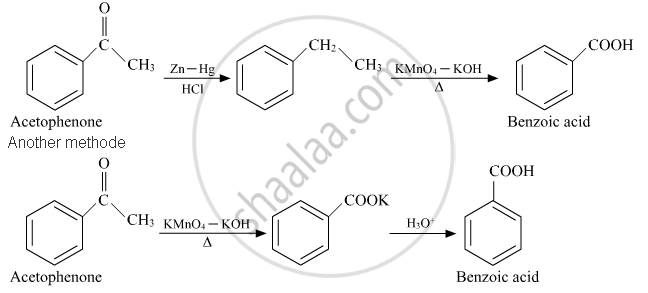
How will you convert the following in not more than two steps:
Acetophenone to Benzoic acid
उत्तर
Acetophenone to Benzoic acid
APPEARS IN
संबंधित प्रश्न
19.5 g of CH2FCOOH is dissolved in 500 g of water. The depression in the freezing point of water observed is 1.0°C. Calculate the van’t Hoff factor and dissociation constant of fluoroacetic acid.
Give reasons for the following
Elevation of the boiling point of 1 M KCl solution is nearly double than that of 1 M sugar solution.
The freezing point depression constant for water is 1.86° K Kg mol-1. If 5 g Na2SO4 is dissolved in 45 g water, the depression in freezing point is 3.64°C. The Vant Hoff factor for Na2SO4 is ______.
We have three aqueous solutions of NaCl labelled as ‘A’, ‘B’ and ‘C’ with concentrations 0.1 M, 0.01 M and 0.001 M, respectively. The value of van’t Hoff factor for these solutions will be in the order ______.
What is the expected each water van't Hoff factor for and K4[F4(CN6)] when it completely dissociated in waters.
Maximum lowering of vapour pressure is observed in the case of ______.
Consider the reaction
\[\begin{bmatrix}\begin{array}{cc}
\phantom{.......}\ce{CH3}\\
\phantom{....}|\\
\ce{CH3CH2CH2 - \overset{⊕}{N} - CH2CH3}\\
\phantom{....}|\\
\phantom{.......}\ce{CH3}
\end{array}\end{bmatrix}\]\[\ce{OH^- ->[Heat] ?}\]
Which of the following is formed in a major amount?
A molecule M associates in a given solvent according to the equation \[\ce{M <=> (M)_n}\]. For a certain concentration of M, the van't Hoff factor was found to be 0.9 and the fraction of associated molecules was 0.2. The value of n is ______.
Why is boiling point of 1 M NaCl solution more than that of 1 M glucose solution?
Calculate Van't Hoff factor for an aqueous solution of K3 [Fe(CN)6] if the degree of dissociation (α) is 0.852. What will be boiling point of this solution if its concentration is 1 molal? (Kb = 0.52 K kg/mol)
