Advertisements
Advertisements
प्रश्न
How will you distinguish between the two black samples, CuO and MnO2 with a chemical test?
उत्तर
Add concentrated hydrochloric acid to both the samples. Only MnO2 releases greenish yellow chlorine gas.
APPEARS IN
संबंधित प्रश्न
Write balanced chemical equations to show The behavior of H2SO4 as an acid when it reacts with Magnesium
Name the gas that is produced in the given cases :
An action of dilute hydrochloride acid on sodium sulphide.
Name an element which reacts with hydrogen to form a compound which is strongly acidic in water.
Give three distinct test [apart from using an indicator] you would carry out with solution of \[\ce{HCl}\] to illustrate the typical properties of an acid.
Name the following:
Gas obtained by treating ferrous sulphide with hydrochloric acid.
Name the following:
A metallic oxide which reacts with hydrochloric acid to give a coloured solution.
Write a fully balanced of the following case :
Red lead is warmed with concentrated hydrochloric acid.
Write observation:
Lead nitrate solution is mixed with dilute hydrochloric acid and heated.
Write an equation for the reaction of hydrochloric acid on manganese (IV) oxide.
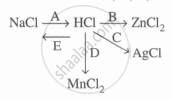
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.