Advertisements
Advertisements
प्रश्न
Identify the polymer given below:
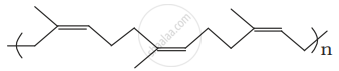
उत्तर
The cis-polyisoprene molecule consists of various chains held together by weak van der Waals interactions and has a coiled structure. Thus, it can be stretched like a spring and exhibits elastic properties.
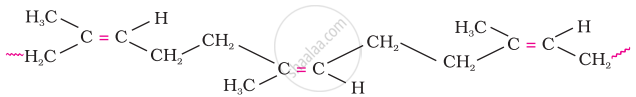
Nautral rubber
APPEARS IN
संबंधित प्रश्न
Write names and chemical formulae of monomers used in preparing Buna-S.
How does the presence of double bonds in rubber molecules influence their structure and reactivity?
Write names and chemical formulae of monomers used in preparing Buna-N.
Complete the following statement by selecting the correct alternative from the choices given:
Natural rubber is a : _________
What is the role of Sulphur in the vulcanization of rubber?
Which of the following are example of synthetic rubber?
(i) Polychloroprene
(ii) Polyacrylonitrile
(iii) Buna-N
(iv) cis-polyisoprene
Vulcanisation makes rubber:
(i) more elastic
(ii) soluble in inorganic solvent
(iii) crystalline
(iv) more stiff
Which of the following is correct regarding the drawbacks of raw rubber.
Which of the following statement is CORRECT regarding the drawbacks of raw rubber?
Match List I with List II.
| List I | List II |
| (Monomer Unit) | (Polymer) |
| (a) Caprolactum | (i) Natural rubber |
| (b) 2-Chloro-1, 3-butadiene | (ii) Buna-N |
| (c) Isoprene | (iii) Nylon-6 |
| (d) Acrylonitrile | (iv) Neoprene |
Choose the correct answer from the options given below:
