Advertisements
Advertisements
प्रश्न
Indicate the number of unpaired electrons in Si.
उत्तर
Silicon (Si):
Atomic number = 14
The electronic configuration of Si is:
1s2 2s2 2p6 3s2 3p2
The orbital picture of Si can be represented as:
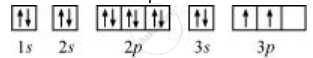
From the orbital picture, silicon has two unpaired electrons.
APPEARS IN
संबंधित प्रश्न
Which atom is indicated by the following configuration?
[He] 2s1
Which atom is indicated by the following configuration?
[Ne] 3s2 3p3
Which atom is indicated by the following configuration?
[Ar] 4s2 3d1.
An atomic orbital has n = 3. What are the possible values of l and ml?
List the quantum numbers (ml and l) of electrons for 3d orbital.
Which of the following orbitals are possible?
1p, 2s, 2p and 3f
Indicate the number of unpaired electrons in a P.
Indicate the number of unpaired electrons in Cr.
Indicate the number of unpaired electrons in Fe.
Indicate the number of unpaired electrons in Kr.
Show the distribution of electrons in oxygen atom (atomic number 8) using orbital diagram.
The atomic number of elements is 17. The number of orbitals containing electron pairs in its valence shell is ______.
The number of 4f electron in the ground state electronic configuration Gd2+ is ______. (Atomic number of Gd = 64)
The correct set of four quantum numbers for the valence electrons of the rubidium atom (Z = 37) is ______.
